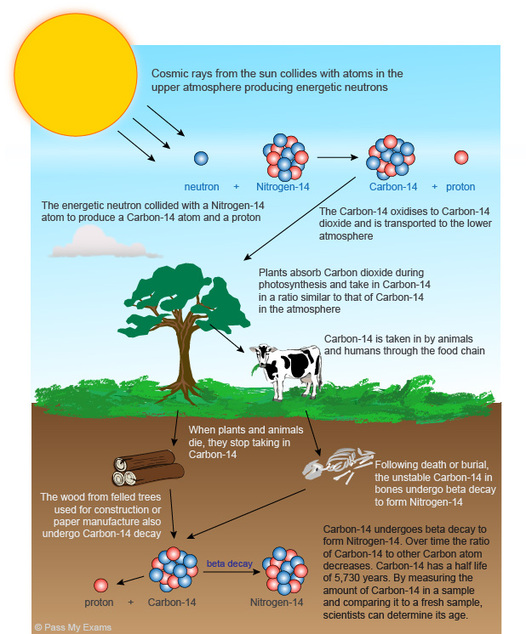
Carbon dating in this manner is really only useful for dating objects less than 60,000 years old. The amount of Carbon-14 remaining after this time is usually too small to measure accurately. The amount of Carbon present in the atmosphere has not always been constant, and this must be allowed for in calculations as well.
Other radio isotopes with longer half lives, such as Uranium-238 have been used in dating geological samples, and the oldest rocks on earth are believed to be about 4 x 10^9 years, with the first fossilised organisms appearing in rocks aged at least 3 billion years ago.
Other radio isotopes with longer half lives, such as Uranium-238 have been used in dating geological samples, and the oldest rocks on earth are believed to be about 4 x 10^9 years, with the first fossilised organisms appearing in rocks aged at least 3 billion years ago.
