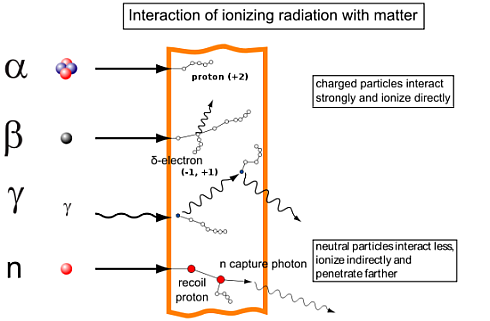
Beta Particle (electron)
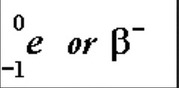
Symbols for a beta negative particle. Note that the first is preferable when balancing equations as it has an atomic number of -1 shown.
What happens in Beta minus decay
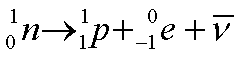
For radioisotopes that have too many neutrons compared to the number of protons, beta minus decay is a way of reducing the number of neutrons. In this process, one neutron is changed to a proton and gives off an electron to balance the charge. The electron given off in this way comes from the nucleus but is identical to an orbital electron. When measured, the energy of the products of the reaction appeared to be less than the parent nucleus, and physicists proposed the existence of the antineutrino to carry off the missing energy (and also the momentum and angular momentum that appeared to be missing from the quantum model-refer to elementary particle section for a brief discussion). Since the antineutrino interacts with matter only via a force called the Weak Nuclear Force, it is much harder to detect.
Beta Decay Equation (electron)
Beta negative decay from element X to element Y. An electron and also an anti neutrino(with close to zero rest mass and zero charge) are given off, along with energy.
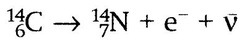
Beta negative decay from Carbon 14 to Nitrogen 14. Atomic number of the left hand side=atomic number ofthe right hand side (6=7+-1). Mass number of the left hand side=mass number of the right hand side (14=14). The antineutrino is considered to be neutral and have no mass when balancing nuclear equations.
Beta Particle (positron)
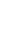
One possible symbol for a positron. In this form, equations can be balanced more easily. Note the atomic number is +1
What happens in Positron Decay
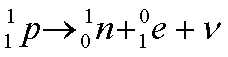
In the case where the nucleus is unstable because there are too many protons for the number of neutrons, one of the protons may change to form a neutron and give off a positron (equivalent to a positively charged electron). A neutrino is postulated to be given off to account for the energy imbalance.
Beta Decay Equation (positron)
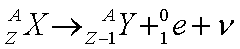
Beta positive or positron decay from element X to element Y. A positron and a neutrino (with close to zero rest mass and zero charge) are given off, along with energy.
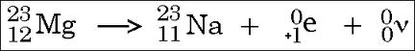
Positron decay from Magnesium 23 to Sodium 23. Atomic mass of the left hand side=atomic number of the right hand side (12 = 11 + 1)
Mass number of the left hand side = mass number of the right hand side (23 = 23) In this example, the neutrino is shown as having atomic and mass numbers of zero for ease of calculations
Mass number of the left hand side = mass number of the right hand side (23 = 23) In this example, the neutrino is shown as having atomic and mass numbers of zero for ease of calculations
Properties of Beta Particles (electrons)
Beta particles are much smaller than alpha particles, with a mass of 9.10938291 × 10^-31 kilograms. They are ejected from the nucleus with a much higher speed, around 2.7 x 10^8 metres/second or around 90% of the speed of light. Because they have a single negative charge and are smaller than alpha particles, they interact less with the atoms of the material through which they are travelling, and are repelled by the electron clouds of the atoms, losing energy in the collisions but not ionising the material to any great degree. They will penetrate further before being slowed and stopped, typically a few metres in air, and will be stopped by a sheet of Aluminium about 1mm thick. They have lower energies than alpha particles, around 1 MeV.