Alpha Particle
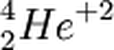
One of the symbols for an alpha particle. Note that it has 2 protons Z=2, 2 neutrons (making a total of 4 nucleons A=4) and a charge of 2+
Alpha Decay Equation
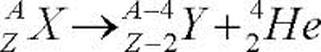
Alpha decay of element X to element Y. Energy is also given off. Atomic and Mass Numbers are Conserved.
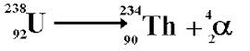
Alpha decay from U-238 to Th-234. This is an alternative symbol for the alpha particle, but is identical in meaning to the Helium nucleus used above. Note the total atomic number on the left=total atomic number on the right (92=90+2) and the total mass number on the left=total mass number on the right (238=234+4).
U-238 is referred to as the Parent nucleus, Th-234 as the Daughter nucleus. This changing from one element into another is called transmutation.
U-238 is referred to as the Parent nucleus, Th-234 as the Daughter nucleus. This changing from one element into another is called transmutation.
Alpha decay takes place among heavy, large nuclei, when the strong nuclear force is no longer able to hold the nucleus together, and the repulsive electric force becomes very large and overpowers it.
The mass of the parent nucleus is greater than the combined mass of the daughter nucleus and the alpha particle. The mass difference appears as Kinetic Energy, which is carried away by the alpha particle and the daughter nucleus. This released energy is called the Disintegration Energy.
The alpha particle is released rather than 4 seperate nucleons because, since the nucleons in the alpha particle are tightly bound, the mass is significantly less than the 4 seperate particles. If the nucleons were seperate, the total mass of the daughter nucleus and the 4 nucleons would be greater than the mass of the parent nucleus, and this would violate the law of the conservation of energy.
The mass of the parent nucleus is greater than the combined mass of the daughter nucleus and the alpha particle. The mass difference appears as Kinetic Energy, which is carried away by the alpha particle and the daughter nucleus. This released energy is called the Disintegration Energy.
The alpha particle is released rather than 4 seperate nucleons because, since the nucleons in the alpha particle are tightly bound, the mass is significantly less than the 4 seperate particles. If the nucleons were seperate, the total mass of the daughter nucleus and the 4 nucleons would be greater than the mass of the parent nucleus, and this would violate the law of the conservation of energy.
Properties of Alpha Particles
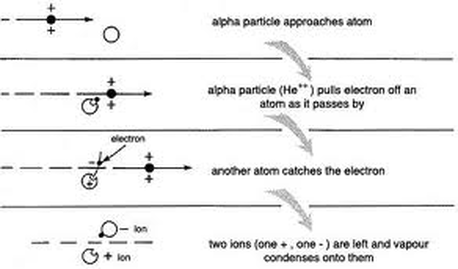
Alpha particles are identical to a Helium nucleus with two protons and two neutrons. Since they have no electrons, this means they have a charge of 2+.This means that as they travel through a medium (eg air), they interact with nearly every atom they encounter. The double positive charge means that they attract electrons from the outer shells of these atoms, causing the atoms to become positively charged ions, and leaving loose electrons behind in their path. The air is said to have become ionised, and the alpha particles are said to have a high ionising ability. Being relatively heavy compared to other radiated particles (mass=4.002602 u, where u=1.66 x 10^-27 kg and is the standard atomic mass, equal to the mass of 1/12 of a Carbon-12 atom), the alpha particle is also relatively slow, with a speed of up to 2 x 10^7 m/s, which is about 10% the speed of light. This makes it even more likely to interact with other atoms and slow down relatively quickly. In air, alpha particles may only travel a few centimetres before they stop, and are easily stopped by a piece of thin card. They are said to have a poor penetrating ability.
The energy of an alpha particle is typically about 5-10MeV, or 8 - 16 x 10^-13 Joules.
The energy of an alpha particle is typically about 5-10MeV, or 8 - 16 x 10^-13 Joules.